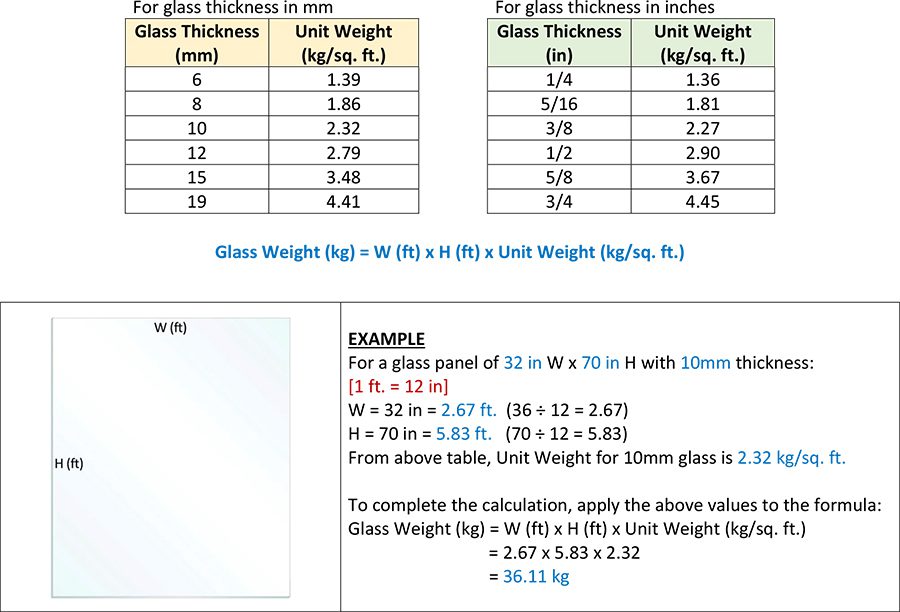
Stainless Steel Grades Comparison
Stainless Steel: 304/304L (Interior Applications)
The material composition of 304 stainless steel is most suitable in applications where it is not introduced to any harsh or severe treatments. The Chromium-nickel alloy of 304 stainless steel makes it somewhat durable and resistant to corrosion and oxidation, though much lower in comparison to 316 Marine Grade and Duplex 2205 stainless steel. It would not be recommended to use 304 or 304L Spigots/Hardware for your frameless glass pool fence as exposure to salt water or chlorinated environments would cause corrosion and oxidation.
Marine Grade Stainless Steel: 316/316L (Exterior Applications)
When exposed to chloride and other chemical-based environments such as salt water, 316 stainless steel shows a high resistance to corrosion, oxidation, and heating as compared to 304 stainless steel. Furthermore, in comparison to 304 stainless steel, it is more durable, easier to fabricate, and requires lower maintenance as it is easier to clean. 316 also holds the advantages of being easier to finish and weld.
Duplex Stainless Steel: 2205 (Highly Corrosive Environments)
Duplex 2205 stainless steel combines most of the advantageous properties of ferritic and austenitic steels, making it one of the highest grades of stainless steel. Due to the duplex nature of the stainless steel alloy, its Pitting and Crevice Corrosion Resistance provides a high resistance to corrosion in most chloride-based environments, giving it a high durability against chloride stress corrosion cracking. Apart from its lowest alloy, all Duplex 2205 stainless steels have much higher tensile strength and yield strength than both 304 and 316 stainless steel combined.
Stainless Steel Cleaning and Maintenance
Learning About and Caring for Stainless Steel
Stainless steel is one of the most durable materials used in architecture, building, and construction. Stainless steel is corrosion-resistant because it forms a thin, protective, passive film on its surface. This film forms spontaneously when chromium in the stainless steel reacts with oxygen in the air. If the film is damaged or removed during fabrication or polishing, it self-repairs quickly as long as the stainless steel surface is clean.
Cleaning and Maintenance
While stainless steel is highly resistant to corrosion, it is not completely impervious. Therefore, regular cleaning is required to preserve the appearance and integrity of its surface. In fact, stainless steel actually thrives with frequent cleaning, and unlike some other materials, it is impossible to “wear out” stainless steel by excessive cleaning.
Regular cleaning with soap or a mild detergent and warm water followed by a clear hot water rinse is usually adequate for household equipment.
For anyone using power wash to clean your pool environment, please be aware that chlorine, dirt, and other particles will likely get sprayed onto the spigots during the cleaning process. To ensure that the residue left on the spigots does not remain and to avoid possible contamination with the stainless steel material, please remember to clean the spigots. You can use soap or a mild detergent and warm water followed by a clear hot water rinse.
Basic Cleaning Tips
The Do’s:
- To avoid water marks, use clean rinse water (ordinary tap water should do). Water spots may be avoided by using an air blower or wiping dry with clean disposable wipes.
- Nylon abrasive pads should be adequate for dealing with most deposits. If more severe treatment is needed to mask scratches or other surface damage, use the finest abrasive possible when removing damage marks. With directional brushed and polished finishes, try to align and blend the new “scratch pattern” with the original finish to achieve the most aesthetic result possible.
- If wire brushes are used, they should be made of a similar or higher-grade stainless steel. Ensure that all abrasives used are free from contamination, especially iron and chloride.
- When cleaning a surface with any chemical preparation or abrasive, a test should first be done on a small, unobtrusive or non-critical surface area in order to verify whether the resulting finish will match the original.
- Under normal use, heavy heat tinting (oxidation) of stainless steel surfaces is unlikely to be encountered. Repeated cleaning with non-abrasive cream cleaners will usually remove burn marks from stainless steel.
- If all of the above suggestions fail, remember that stainless steel can be mechanically polished or electro-polished by specialists on-site. Stainless steel is homogeneous and does not rely on surface plating for its corrosion-resistant properties.
The Don’ts:
- AVOID ALL CONTACT WITH PRODUCTS CONTAINING HYDROCHLORIC ACID SUCH AS BLEACH as they can stain and damage the surface of stainless steel appliances. Agents containing chloride, such as hydrochloric acid, must be avoided since there is an obvious risk of pitting corrosion. Some tile cleaners, toilet bowl cleaners, and pool chemicals contain hydrochloric acid.
- Do not leave stainless steel objects to soak for long periods in chlorine solutions (e.g., overnight). Long-term exposure to table salt and salt and vinegar mixes can damage stainless steel.
- Avoid the use of metal scourers and coarse abrasives or abrasive powders unless absolutely necessary. The fine particles present in steel wool can get lodged in the surface and will eventually rust, giving the appearance that the stainless steel itself is rusting.
- Do not use metal scourers or brushes with metal bristles.
- Do not allow ordinary steel to come into contact with stainless steel under damp conditions.
- Avoid contact with aggressive chemicals. Of particular concern is exposure to chlorine, commonly used to sanitize equipment, as well as hydrochloric acid, used in certain cleaning agents and process liquids.
Ref.:
http://www.julien.ca
http://www.nickelinstitute.org
http://www.outokumpu.com
http://www.hunker.com
Shower Door Hardware Maintenance
The Do’s:
- To maintain the hardware, the best method is to dry it with a clean, soft towel on a regular basis (i.e., at least once daily).
- If you discover mineral deposits, use a mild liquid soap and mix with warm water. Then, be sure to use a soft, non-abrasive cloth (e.g., a microfibre cloth) to clean the hardware. Rinse thoroughly with clean, warm water and dry the hardware.
- If the hardware requires lubrication, apply a drop of general-purpose lubricating oil (“household oil”) on the white nylon washer on the pivoting portion of the body of the hardware. Should the hardware be disassembled, use white lithium grease to lubricate the interior mechanism upon re-assembly.
The Don’ts:
- NEVER use an abrasive cleaner on the hardware. The damages to the hardware will be irreparable if it comes into contact with harsh, abrasive chemicals. The reason is that the hardware is coated with a finish (either plated or painted).
- Do NOT use spray lubricant on the hardware. If lubrication is required, please refer to our instructions above in The Do’s section.
- Do NOT clean the hardware with any chemical liquids.
- Keep hardware away from any kind of acidic and corrosive material.
- Do NOT install the hardware in a corrosive environment.
Anodized Aluminum
What is Anodizing?
Anodizing is an electrochemical conversion process in existence since the 1930s. Several metals are capable of being anodized, including aluminum, magnesium, titanium, and tantalum. Anodized aluminum is used in many applications due to its low cost, aesthetic qualities, and ideal mechanical properties.
Unlike most protective coatings, anodizing permanently changes the outer structure of the metal. When aluminum is exposed to air, it naturally develops a thin aluminum oxide film that seals the aluminum from further oxidation. The anodizing process makes the oxidized surface much thicker, up to several thousandths of an inch thick. The hardness of the anodized aluminum oxide coating rivals that of a diamond, enhancing the abrasion resistance of the aluminum. The added depth of the oxide layer improves the corrosion resistance of the aluminum, while making cleaning of the surface easier. The porous nature of particular types of anodizing makes it possible to dye the aluminum in a variety of colours, making it more attractive.
Anodizing typically ranges up to 5mm thickness. The three most common variations of aluminum anodizing include chromic (type I), sulfuric (type II), and hard (type III).
Aluminum Anodizing Process
- Aluminum part is dipped in a hot tank containing a soak cleaning agent to remove all surface dirt.
- The part is rinsed to avoid contaminating the solution in subsequent tanks.
- The next tank de-oxidizes the part with an acid solution (chromic, sulfuric, nitric, or phosphoric), removing the thin, non-uniform aluminum oxide surface.
- Again, the part is rinsed to avoid tank contamination.
- Etching is performed by suspending the part in a tank containing a sodium hydroxide solution. Etching removes the natural shine of the aluminum and provides a soft, matte, textured appearance.
- The part is suspended in the anodizing tank, which contains a diluted acid and water mixture that is capable of permitting electrical current flow. The type of acid, percent solution, and temperature are all critical parameters dependent on the desired finish and colour. The negative side of the electrical circuit is connected to the part and the positive side of the circuit is connected to one or more “cathodes” that introduces electricity into the tank. The anodizing tank process, under normal conditions, takes less than an hour.
- To add colour (dye), the part is dipped in a tank with a diluted, water soluble, organic dye. Each dye varies in the length of time and temperature for this immersion.
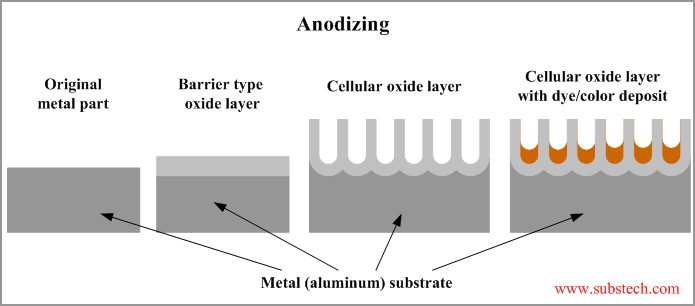
Why Anodize?
Anodizing is a highly effective and desirable means of finishing aluminum. Some of the primary advantages of anodizing include:
Durability – Most anodized parts experience no wear from handling, installation, usage, and maintenance.
Adhesion – Anodizing is part of the aluminum for total bonding and unmatched adhesion.
Colour – Anodized parts maintain good colour stability when exposed to ultraviolet rays, do not have an applied coating that is subject to chipping or peeling, and have a repeatable colouring process.
Quality of original finish – Parts are not subject to marking from the original anodizing process.
Maintenance – Mild soap-and-water cleaning usually will restore an anodized profile to its original appearance.
Aesthetics – Anodizing offers a large number of gloss and colour alternatives, while allowing the metallic appearance of the extruded aluminum to show through.
Cost – Anodizing is very cost-effective compared to other finishing methods. In addition to low processing and maintenance costs, durability minimizes replacement costs.
Environment, health, and safety – Anodizing is favourable towards current governmental regulations because it is one of the most environmentally-friendly industrial processes and is typically not harmful to human health. An anodized finish is chemically stable, will not decompose, is non-toxic, and is heat-resistant to the melting point of aluminum. Since the anodizing process is a reinforcement of a naturally-occurring oxide process, it is non-hazardous and produces no harmful or dangerous by-products. Chemical baths used in the anodizing process are often reclaimed, recycled, and reused.
Ref.: http://www.defelsko.com
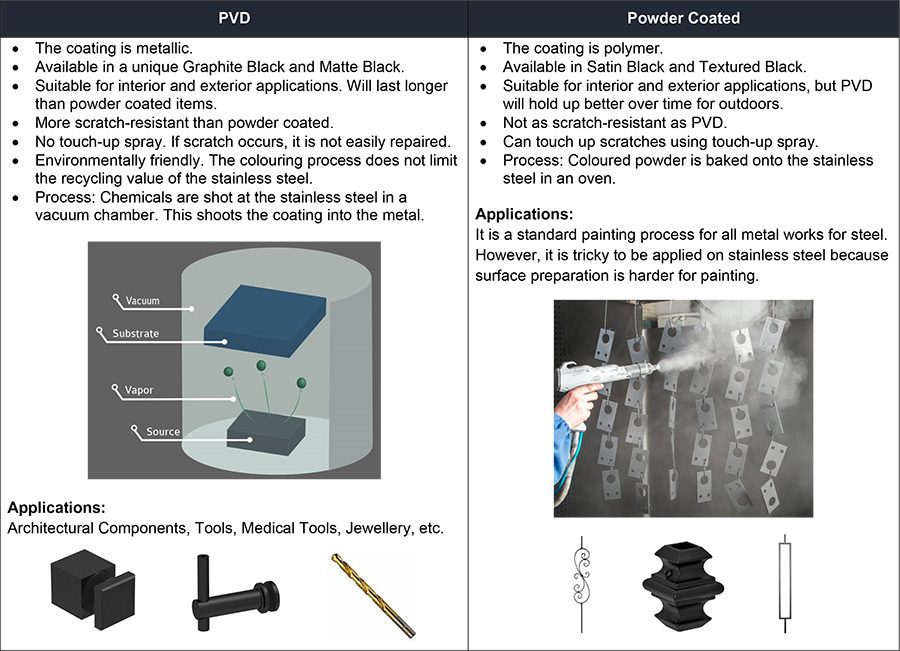
PVD vs. Powder Coated Items – What is the difference?
Uses of Everbrite vs. ProtectaClear
Please check our Everbrite & ProtectaClear Guide comparing the uses of the two types of coatings. Instructions have been included as well. For Quick Tips on “Which Coating Do I Need?”, download: English | français.